Goal 2 - pH Scale Notes
pH and the pH Scale
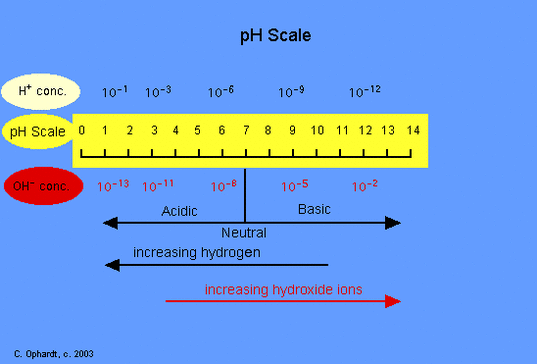
The pH scale ranges from 0 to 14.
The pH scale measures the acidity or basicity of a solution. A pH of 7 means it is a neutral solution.
pH
Pure water has a pH of 7.
A pH of less than 7 means the solution is acidic.
A pH of more than 7 means the solution is basic.
The less pH, the more acidic the solution is.
The more pH, the more basic the solution is.
Acids
An acid is any substance that forms hydrogen ions (H+) in water.
Acidic solutions contain an abundance of H+ ions and have a pH below 7.
Acidic solution turn blue litmus paper red.
Bases
A base is any substance that forms hydroxide ions (OH-) in water.
Basic solutions have an abundance of OH- ions and have a pH of greater than 7.
Basic solutions turn red litmus paper blue.
Examples
Acids: lemons (pH = 2), tomato (pH = 4), milk (pH = 6)
Bases: egg (pH=8), antacid (pH=10), household ammonia (pH=11), drain cleaner (pH=13)
The pH scale ranges from 0 to 14.
The pH scale measures the acidity or basicity of a solution. A pH of 7 means it is a neutral solution.
pH
Pure water has a pH of 7.
A pH of less than 7 means the solution is acidic.
A pH of more than 7 means the solution is basic.
The less pH, the more acidic the solution is.
The more pH, the more basic the solution is.
Acids
An acid is any substance that forms hydrogen ions (H+) in water.
Acidic solutions contain an abundance of H+ ions and have a pH below 7.
Acidic solution turn blue litmus paper red.
Bases
A base is any substance that forms hydroxide ions (OH-) in water.
Basic solutions have an abundance of OH- ions and have a pH of greater than 7.
Basic solutions turn red litmus paper blue.
Examples
Acids: lemons (pH = 2), tomato (pH = 4), milk (pH = 6)
Bases: egg (pH=8), antacid (pH=10), household ammonia (pH=11), drain cleaner (pH=13)

